 Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron. The use of infrared spectroscopy began in the 1950s by Wilbur Kaye. An absorption spectrum occurs when light passes through a cold, dilute gas and atoms in the gas absorb at characteristic frequencies since the re-emitted light. A spectrum obtained from electromagnetic energy transmitted throughout a gas or any solid is referred to as an absorption spectrum. 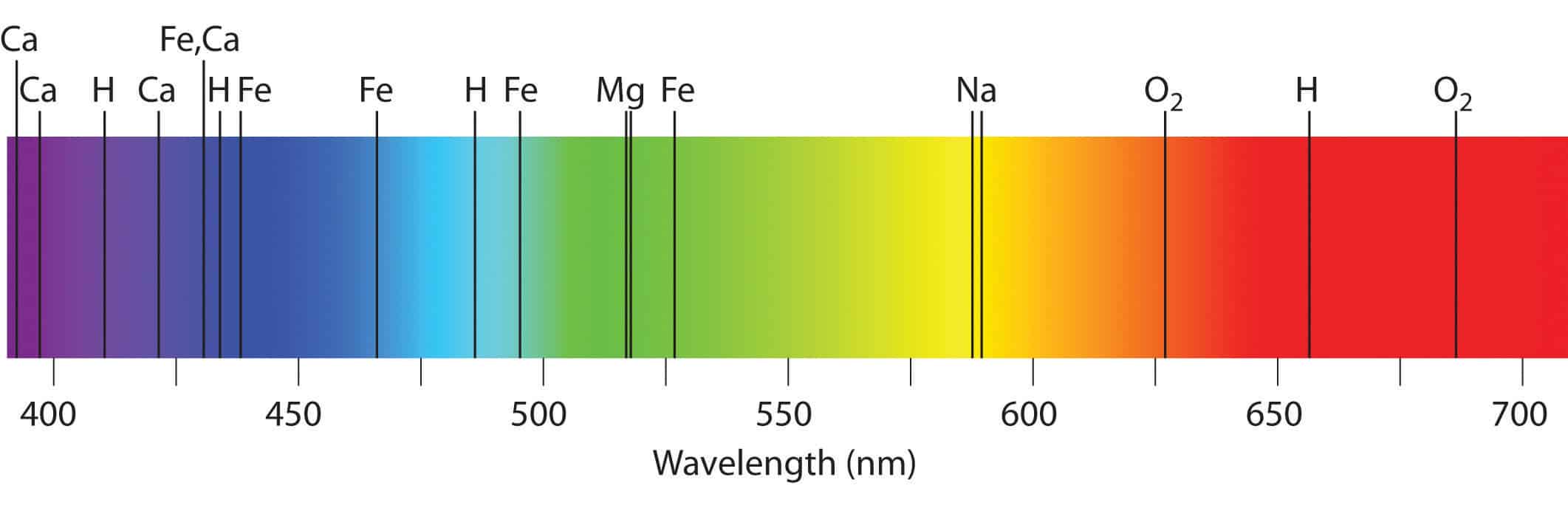
Figure 1.4.3: German chemist Robert Bunsen (1811 - 1899). It can also be used for both qualitative and quantitative analysis of complex mixtures of similar compounds. Although Bunsen and Kirchoff took a large step in defining the technique of atomic absorption spectroscopy (AAS), it was not widely utilized as an analytical technique except in the field of astronomy due to many practical difficulties. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. IR spectroscopy is very useful in the identification and structure analysis of a variety of substances, including both organic and inorganic compounds. UV-Vis spectroscopy detects energy from the excitation of electrons at different wavelengths to determine where maximum absorbance occurs. In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
